A. Introduction
Hydrogen, the simplest and lightest element, reigns as the most abundant in the universe, constituting approximately 75% of its elemental mass[1][2] . Created during the Big Bang, hydrogen atoms formed just minutes after the universe’s inception, making them the building blocks of stars, planets, and interstellar gas[2] . This primordial element fuels the nuclear fusion in stars like our Sun, which is primarily composed of hydrogen and helium . Even gas giants such as Jupiter and Saturn mirror this cosmic recipe, with their atmospheres dominated by hydrogen .
Atomic Number : 1, indicating one proton in its nucleus
Relative Atomic Mass : 1.008
State at 20°C : Gas
Electron Configuration : 1s¹ (single electron in the 1s orbital)
Isotopes : Hydrogen-1 (¹H, protium) and deuterium (²H) are notable
CAS Number : 133-74-0
Unique Property : Lacks core electrons, so it only appears in the valence band in XPS analysis
[1]
Hydrogen on Earth: Hidden in Plain Sight
Despite its universal prevalence, elemental hydrogen (H₂) is scarce on Earth. Here, it exists almost exclusively in compounds: water (H₂O), hydrocarbons, and organic matter[2] . Water, covering 71% of Earth’s surface, is its most familiar reservoir. Yet isolating pure hydrogen is challenging. Its high reactivity causes it to bond readily with other elements, and when released as a gas, it disperses quickly due to its low density .
B. The Paradox of Abundance and Scarcity
Elemental hydrogen’s rarity stems from its physical properties. As the smallest molecule, it escapes Earth’s gravity easily, lingering in the atmosphere at just 0.5–1.0 parts per million[2] . When concentrated, its flammability becomes a hazard—mixing with oxygen in air creates explosive mixtures, complicating storage and transport . These challenges have hindered its adoption as a clean energy carrier, despite its potential.
1. Ionization Energy (1312.05 kJ/mol)
Hydrogen has an extremely high first ionization energy compared to most elements.The electron in a hydrogen atom occupies the 1s orbital, the smallest and most tightly bound orbital, resulting in weak intermolecular van der Waals forces between H₂ molecules .
This means that it requires significant energy to remove its single electron, making atomic hydrogen (H) highly reactive and unstable under Earth’s ambient conditions.Instead of existing as free atoms, hydrogen preferentially forms stable covalent bonds with other elements (e.g., H₂O, CH₄)
2. Bond Enthalpies
Hydrogen forms exceptionally strong bonds with other elements, as shown in the table[1]:
- H–H bond (435.9 kJ/mol) : While strong, this bond is weaker than bonds formed with oxygen (H–O: 462.8 kJ/mol in H₂O) or carbon (C–H: ~413–415.5 kJ/mol in CH₄).
- These stronger bonds in compounds like water, methane, and ammonia trap hydrogen in stable molecules, reducing its availability as free H₂ gas
3. Planetary Conditions :
Earth’s gravity is too weak to retain lightweight H₂ gas, allowing it to escape into space. Oxygen-rich environments (like Earth’s atmosphere) promote oxidation of hydrogen into water, further locking it into compound.
C. Hydrogen Cycle
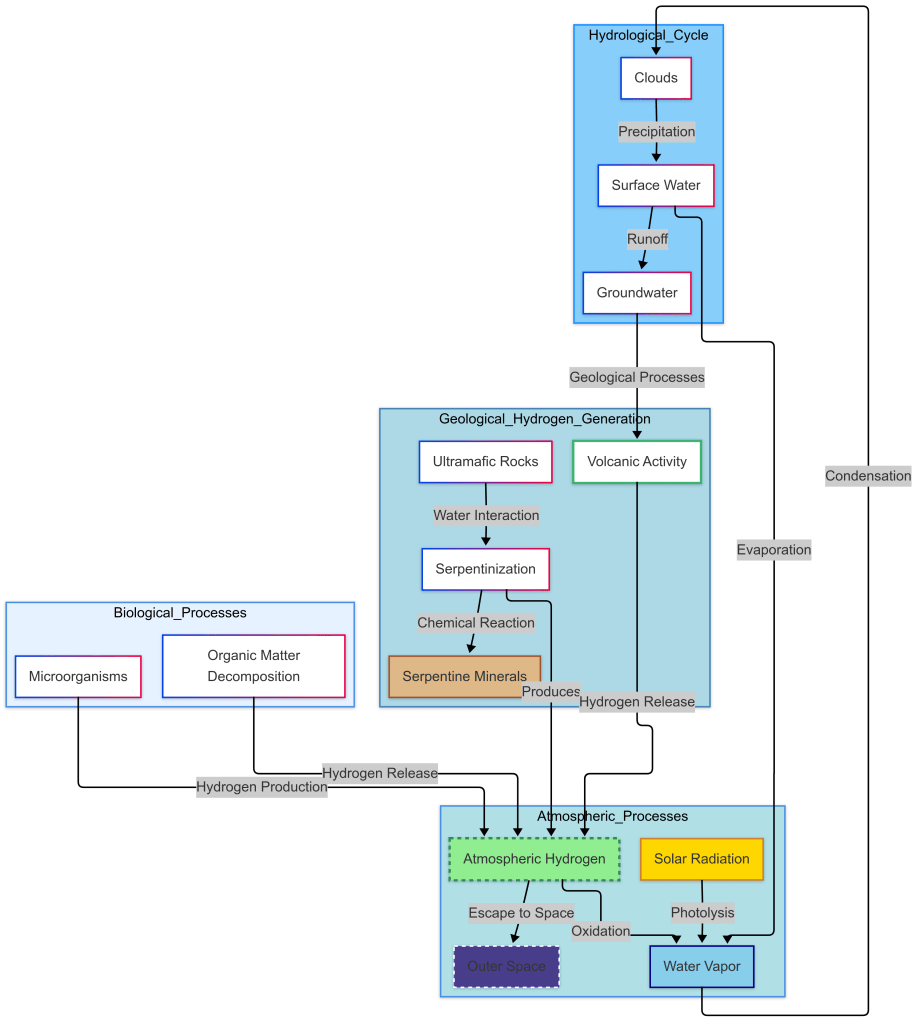
The hydrogen cycle plays a critical role in maintaining Earth’s water balance and supporting ecosystems by facilitating hydrogen exchanges between biotic and abiotic systems[4] . It integrates with the hydrological cycle through processes like evaporation and precipitation, ensuring water availability for living organisms . Industrially, hydrogen is vital for applications such as ammonia production and clean energy , while its geological generation via serpentinization and volcanic activity contributes to atmospheric hydrogen levels[4] . However, climate change accelerates the hydrological cycle, intensifying extreme weather events , underscoring the interconnectedness of hydrogen dynamics and planetary health [3]
Hydrogen Leakage and Atmospheric Chemistry :
While hydrogen itself is not a greenhouse gas, its leakage can indirectly amplify climate impacts. Atmospheric hydrogen interacts with hydroxyl radicals (OH⁻), reducing their capacity to break down methane (CH₄), a potent greenhouse gas . This prolongs methane’s atmospheric lifetime, enhancing its warming effect .[5]
Sources:
[1] https://periodic-table.rsc.org/element/1/hydrogen
[2] https://www.acs.org/molecule-of-the-week/archive/h/hydrogen.html
[3]https://www.nature.com/articles/s43247-025-02141-3
[4] https://en.wikipedia.org/wiki/Hydrogen_cycle
[5] https://acp.copernicus.org/articles/22/9349/2022/

Leave a comment